Our medical services use preventative treatments to ensure patients have the tools they need to maintain good health. With several different primary care options, we help patients improve their physical condition and provide education that improves their long-term outlook.
Looking your best plays a major role in feeling your best. Our medical spa in San Antonio offers a range of aesthetic treatments capable of enhancing your whole body—rejuvenating your appearance and boosting your confidence.
Feeling your best goes beyond physical health and disease prevention. Your mental and sexual health are just as important to your overall wellbeing. The providers at Marcos Medical Care know this—and they can help both men and women with treatments designed to improve their love lives.
Your Health & Wellness is Our Priority
Marcos Medical puts patients at the center of care from the moment they walk in the door.
Our internal medicine practice and medical spa in San Antonio use a combination of consistent learning and cutting-edge technology to provide the most advanced treatments available. But more importantly, we collaborate with patients to provide integrated care that fits their needs and achieves their goals.

Monthly Special
Aesthetics Fiesta Sale 💃
Join us at Marcos Medical Care for an exclusive celebration of beauty and wellness! For a limited time only, we’re offering an extraordinary 25% off all our aesthetic treatments. Dive into the Fiesta of rejuvenation and pampering with our diverse range of services designed to enhance your natural beauty and well-being.
Our Aesthetics Fiesta Sale includes:
- Body Contouring: Sculpt and redefine your body with our advanced contouring treatments.
- Laser Hair Removal: Achieve smooth, hair-free skin with our precise laser technology.
- Facials & Hydrafacials: Refresh and revitalize your complexion with our customized facial treatments.
- RF Microneedling: Experience the transformative effects of RF Microneedling for skin rejuvenation.
- Hair Restoration: Restore your hair’s natural thickness and growth with our effective treatments.
- Skin Resurfacing: Reveal younger, healthier-looking skin with our cutting-edge skin resurfacing techniques.
This is the perfect opportunity to indulge in some self-care and embrace the festive spirit of our Aesthetics Fiesta!

Who is Marcos Medical?
Starting over 20 years ago, Marcos Medical Care has provided excellent long-term outcomes to patients across North Central San Antonio and beyond
Our patients are our purpose, and they’re the reason that we strive to provide the most advanced care available. The heart of our medical spa in San Antonio is our team of extraordinary provers- who use experience and compassion to improve patients’ health and wellness.
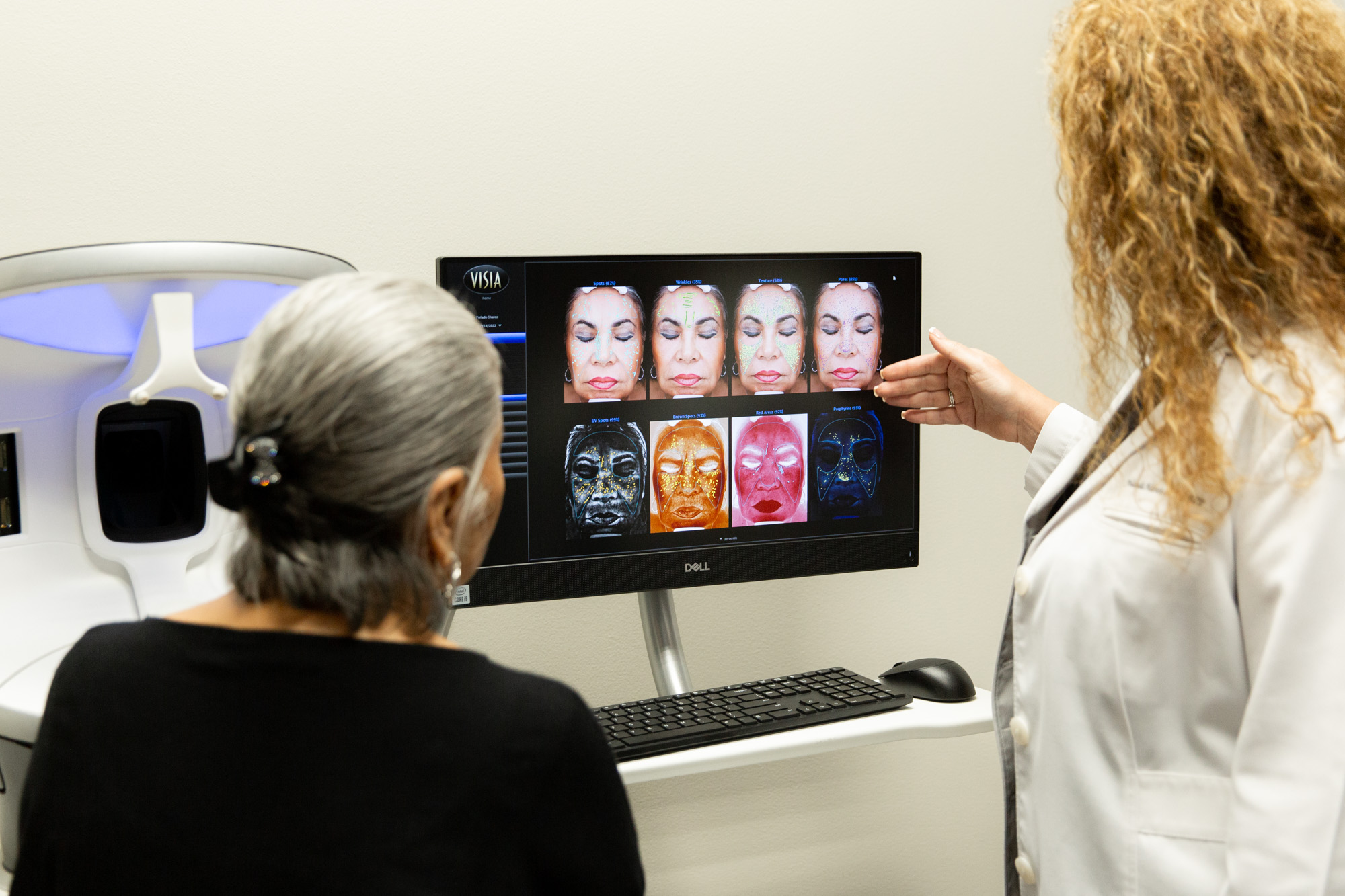
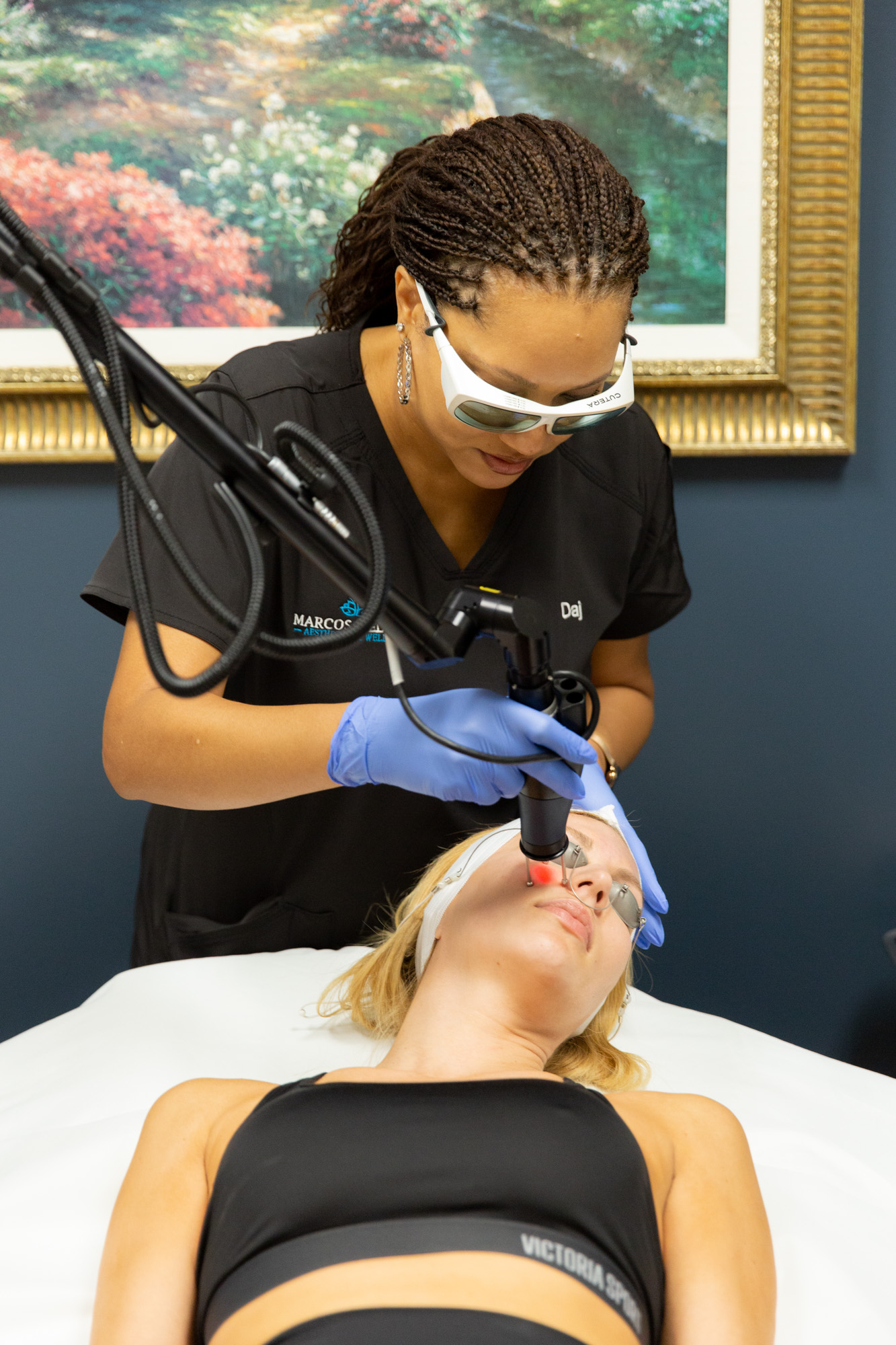
Benefits of Visiting Marcos Medical
Quality
Marcos Medical has earned several honors as a top care center and medical spa in Stone Oak and the Greater San Antonio area. Our health plans and evidence-based medicine show our commitment to providing excellent patient care.
Caring Providers
Our providers are our biggest differentiator. Each and every one of them deeply cares for the health and well-being of our patients, and they'll work tirelessly to help you reach your goals.
Advanced Treatments
Our team believes in constant improvement and lifelong learning. Investing in the latest, most proven treatments helps patients get the exact results they're looking for.
Collaboration
Your visit to Marcos Medical is all about you. We don't just practice care for patients—we listen to their issues and work together to find a treatment that fits their needs, wants, and goals.